Hypertrophic cardiomyopathy occurs when the size of heart muscle cells increase (aka: hypertrophy). Genetic mutations in DNA that codes for heart muscle cell proteins are responsible for the development of hypertrophic cardiomyopathy. Most of these mutations are in DNA that code for sarcomere proteins (ie: myosin, actin, troponin, etc.). The mutated proteins cause decreased contractile function. As a result, the muscle cell hypertrophies (enlarges) in an attempt to overcome the decreased contractility. The result is a disorganized pattern of muscle cell fibers with intervening fibrosis (ie: scar tissue).
Signs and Symptoms
Since the myocardium is hypertrophied there is less ventricular compliance (ie: the heart becomes stiff). This stiffness decreases the filling capacity of the ventricle. The result is diastolic dysfunction, or a decreased ability of the heart to fill during its relaxation phase. High diastolic pressures occur leading to the back-up of blood into the left atrium, pulmonary veins, and pulmonary capillaries. Excess fluid in the pulmonary capillaries causes pulmonary edema with resultant shortness of breath and exercise intolerance.
In addition, angina (chest pain) can occur even without co-existing coronary artery disease because the increased muscle mass of the ventricle results in a higher oxygen demand. Under strenuous conditions the hypertrophied muscle cannot get enough oxygen, which causes chest pain.
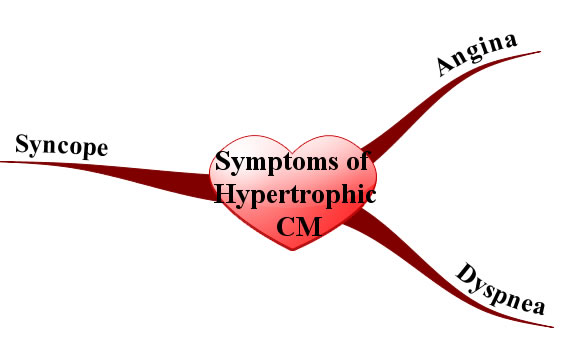
Physical exam can reveal an S4 gallop (aka: atrial gallop), which is caused by the atrium forcing blood into a stiff left ventricle during the "atrial kick" at the end of diastole.
Murmurs can also be heard, usually mitral regurgitation and a systolic outflow obstruction murmur. Mitral regurgitation occurs because the hypertrophied ventricular septum acts as a barrier to blood flow into the aorta. As a result, during systole blood will flow backwards through the mitral valve into the left atrium. Blood flowing across the septal barrier into the aorta will create an obstruction murmur. The obstruction murmur worsens with valsalva, which distinguishes it from the murmur of aortic stenosis.
Diagnosis
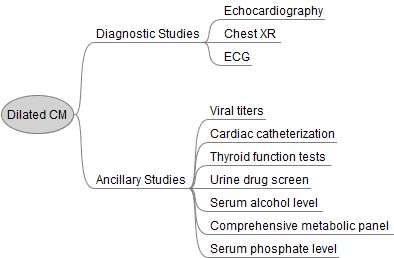
The work-up is very similar to dilated cardiomyopathy, except ancillary studies are usually not helpful. Echocardiography (ie: ultrasound of the heart) is the gold standard and will show the hypertrophic myocardium. ECG will often reveal left ventricular hypertrophy and left atrial hypertrophy. Arrhythmias may sometimes be observed on ECG as well. Prominent Q-waves can be seen in the lateral leads (ie: V4-V6) and inferior leads (II, III, aVF); this is the result of greater depolarization of the hypertrophied septum (remember depolarization of the septum starts on the left side and moves rightward creating a downward deflection in leads on the opposite side of the body, before the left ventricle "overpowers" the ECG findings).
Treatment
β-blockers are the mainstay of treatment. They decrease the heart rate and allow increased diastolic filling times, which leads to decreased outflow obstruction; they also decrease myocardial oxygen demand leading to decreased anginal symptoms.
Prevention of fatal arrhythmias is important in hypertrophic cardiomyopathy. Medical management of arrhythmias is accomplished with amiodarone and/or disopyramide. In some patients, strong consideration should be given to an implantable cardiac defibrillator, especially those at high risk of sudden death. Surgery with partial myomectomy to remove some of the hypertrophied muscle can also be done if the patient is unresponsive to medical management.
Since hypertrophic cardiomyopathy is caused by genetic mutations, genetic counseling should be offered to children of affected parents. First degree relatives should undergo screening with echocardiography as well.
Unlike dilated cardiomyopathy, diuretics should be used sparingly because they can worsen outflow obstruction by causing decreased venous return to the left ventricle. Digoxin is also contraindicated because it can worsen outflow obstruction. When considering treatment options it is important to remember that the problem in hypertrophic cardiomyopathy is diastolic, not systolic dysfunction.
Prognosis depends on the type and severity of the genetic mutation involved. Some mutations result in minimal morbidity and a normal life span, whereas others can cause significant heart failure symptoms. Overall mortality is roughly 5% per year secondary to ventricular fibrillation; therefore, even minimally symptomatic patients must be monitored closely.
Overview
The cause of hypertrophic cardiomyopathy is genetic. Diagnosis is made with echocardiography (ie: ultrasound of the heart). Treatment is generally with beta blockers, amiodarone, implantable cardiac defibrillators (ICD), and myomectomy in select patients. All 1st degree relatives should be offered genetic counseling and undergo screening echocardiography. Prognosis is variable and depends on the mutation type.
Related Articles
- Dilated Cardiomyopathy: Poor Pump, S3, and Crackles
- Restrictive Cardiomyopathy: Amyloid, Diastolic Dysfunction, and Kussmaul’s Sign
- Cardiac Output: Pump, Pump, Squeeze
- AV Heart Block: PR, QRS, and Mobitz
References and Resources
- Bos JM, Ommen SR, Ackerman MJ. Genetics of hypertrophic cardiomyopathy: one, two, or more diseases? Curr Opin Cardiol. 2007 May;22(3):193-9.
- Bo CY, López B, Coelho-Filho OR, et al. Myocardial fibrosis as an early manifestation of hypertrophic cardiomyopathy. N Engl J Med. 2010 Aug 5;363(6):552-63.
- Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. Seventh Edition. Philadelphia: Elsevier Saunders, 2004.
- Lilly LS, et al. Pathophysiology of Heart Disease: An Introduction to Cardiovascular Disease. Seventh Edition. Lippincott Williams and Wilkins, 2006.
- Flynn JA. Oxford American Handbook of Clinical Medicine (Oxford American Handbooks of Medicine). First Edition. Oxford University Press, 2007.